Structural Biology looks at proteins and nucleic acids at a molecular level, unlocking secrets of structure, function and interactions, and providing new avenues for medical research. How are the amino acids or nucleic acids arranged in three dimensions at atomic resolution, and how does this relate to their function? What does a binding site look like, and how do ligands become attached? What conformational changes result after ligand binding? How do macromolecular complexes form? What are the effects of point mutations in particular diseases? What are potential sites for drug action, and how could such drugs be designed? Where does artificial intelligence fit into the evolving world of structural studies? All these and other questions are answered by Structural Biology.
Revolutionary Biology Part 2: The history of structural biology from NDM Oxford.
Whilst structural biology was once a niche area, it is now accessible to life scientists across all disciplines, and should be part of the toolbox for all researchers.
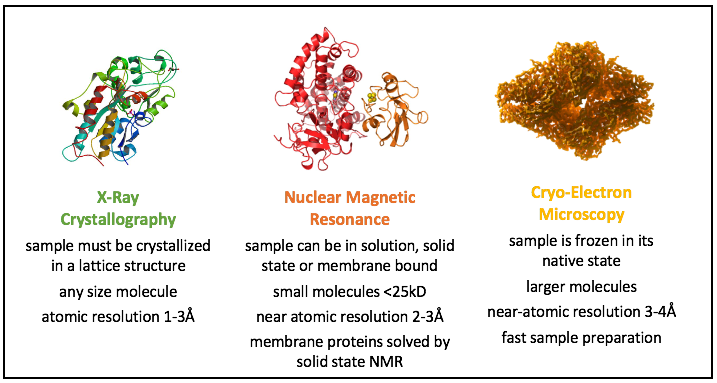
In recent years we have seen a revolution in structural biology. Macromolecular crystallography has become a mature technique that permits structures to be obtained rapidly after sample crystallisation, results that can be complemented by small angle solution X-ray scattering. NMR has emerged as a particularly useful technique to examine macromolecular dynamics in solution and membrane proteins in solid state. Cryo electron microscopy (cryo-EM) has exploded with the advent of direct detectors that permit near atomic resolution on macromolecular complexes. An array of biophysical methods provide additional molecular information. And in recent years, artificial intelligence has changed the way we identify protein targets for structural studies.
The three main tools used for protein structure determination, all available to access through the Instruct catalogue.